Einecs 223-785-3
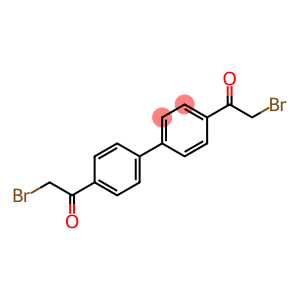
1,1'-[1,1'-biphenyl]-4,4'-diylbis[2-bromoethan-1-one]
CAS: 4072-67-7
Molecular Formula: C16H12Br2O2
Einecs 223-785-3 - Names and Identifiers
| Name | 1,1'-[1,1'-biphenyl]-4,4'-diylbis[2-bromoethan-1-one] |
| Synonyms | Einecs 223-785-3 4,4'-Bis(bromoacetyl)biphenyl 4,4'-Bis(2-bromoacetyl)biphenyl 1,1'-biphenyl-4,4'-diylbis(2-bromoethanone) 1,1'-(1,1'-Biphenyl-4,4'-diyl)bis(2-bromoethanone) Ethanone, 1,1'-(1,1'-biphenyl)-4,4'-diylbis(2-bromo- 2-Bromo-1-[4-[4-(2-bromoacetyl)phenyl]phenyl]ethanone 1,1'-[1,1'-biphenyl]-4,4'-diylbis[2-bromoethan-1-one] 1,1'-(1,1'-Biphenyl)-4,4'-diylbis(2-bromoethan-1-one) 4,4'-Bis(2-bromoacetyl)biphenyl 1,1'-(1,1'-Biphenyl)-4,4'-diylbis(2-bromoethan-1-one) |
| CAS | 4072-67-7 |
| EINECS | 223-785-3 |
| InChI | InChI=1/C16H12Br2O2/c17-9-15(19)13-5-1-11(2-6-13)12-3-7-14(8-4-12)16(20)10-18/h1-8H,9-10H2 |
Einecs 223-785-3 - Physico-chemical Properties
| Molecular Formula | C16H12Br2O2 |
| Molar Mass | 396.07 |
| Density | 1.622±0.06 g/cm3(Predicted) |
| Melting Point | 226-227 °C(Solv: benzene (71-43-2)) |
| Boling Point | 464.8±30.0 °C(Predicted) |
| Flash Point | 121.3°C |
| Vapor Presure | 8.12E-09mmHg at 25°C |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | 1.625 |
Einecs 223-785-3 - Risk and Safety
| HS Code | 29147000 |
Einecs 223-785-3 - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| overview | 4,4 '-bis (2-bromoacetyl) biphenyl is a key intermediate in the synthesis of the anti-hepatitis C drug dhathavir. Dhaka Tamivir (Daclatasvir) is a new hepatitis C drug developed by Bristol-Myers Squibb in the United States. On July 24, 2015, the US FDA approved it for the treatment of chronic hepatitis C. Its trade name is Daklinza. It is an NS5A inhibitor and is suitable for adults infected with genes 1, 2, 3 and 4. Its chemical name is N,N'-[[1,1 '-biphenyl]-4,4'-diyl bis [1H-imidazol-5, 2-diyl-(2S)-2, 1-pyrrolidinyl [(1S)-1-(1-methylethyl) -2-oxo-2, 1-ethanediyl]] bis carbamate C, c'-dimethyl ester, the synthesis of 4,4 '-bis (2-bromoacetyl) biphenyl is reported in the literature. Biphenyl and bromoacetyl bromide are used as substrates and CS is used as the reaction solvent. Friedel acylation reaction occurs under the action of AlCl to obtain the product 4,4'-bis (2-bromoacetyl) biphenyl. |
| application | 4,4 '-bis (2-bromoacetyl) biphenyl is an organic synthesis intermediate and a pharmaceutical intermediate, which can be used as a laboratory research and development process and a chemical medicine research and development process. |
| Preparation | 4,4 '-bis (2-bromoacetyl) biphenyl The preparation method includes the following steps:(1) At the reaction temperature of -10 ℃ ~ 30 ℃, the catalyst and chlorinated alkane solvent are added to the reaction bottle to mix, the system is cooled to -10~30 ℃, the bromoacetyl bromide is added to the upward system dropwise, heat preservation and stirring for 10~30 minutes. The chlorinated alkane solution containing biphenyl is added dropwise to cause the Friedel-Crafts acylation reaction between bromoacetyl bromide and biphenyl to obtain the first mixed system. The chlorinated alkane solvent includes, but is not limited to: dichloromethane, 1, 2-dichloroethane, chloroform and/or carbon tetrachloride; the catalyst includes, but is not limited to: aluminum trichloride, zinc chloride and/or ferric chloride. (2) heating to room temperature and stirring continuously, adding anti-solvent to the first mixed system, and continuing the reaction at the reaction temperature of 20 ℃ ~ 60 ℃ for 0.5~8 hours to obtain a second mixed system containing 4,4 '-bis (2-bromoacetyl) biphenyl; the anti-solvent is selected from C3 ~ C10 alkanes, ethers and esters; C3 ~ C10 alkanes are selected from: n-hexane, n-heptane, cyclohexane, methylcyclohexane, pentane, octane, nonane, decane; ethers are selected from: petroleum ether, methyl tert-butyl ether, ether, ethylene glycol dimethyl ether, tetrahydrofuran, 2-methyltetrahydrofuran, isopropyl ether; esters are selected from: ethyl acetate, butyl acetate, isopropyl acetate; preferably n-hexane, cyclohexane, n-heptane, petroleum Ether; better n-heptane, cyclohexane. (3) Control the temperature of the second mixed system containing 4,4 '-bis (2-bromoacetyl) biphenyl at -5~25 ℃, stir for 2~5 hours, and filter to obtain 4,4'-bis (2-bromoacetyl) biphenyl crude product; the obtained crude product is beaten in water and dichloromethane solution at room temperature, filtered, and dried to obtain high-quality products 4,4 '-bis (2-bromoacetyl) biphenyl. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 4,4'-Bis(2-bromoacetyl)biphenyl Visit Supplier Webpage Request for quotationCAS: 4072-67-7
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 1,1'-([1,1'-Biphenyl]-4,4'-diyl)bis(2-bromoethanone) Visit Supplier Webpage Request for quotationCAS: 4072-67-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4,4'-Bis(2-bromoacetyl)biphenyl Request for quotation
CAS: 4072-67-7
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 4072-67-7
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: Isavuconazole Impurity 128 Request for quotation
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: 1,1'-[1,1'-biphenyl]-4,4'-diylbis[2-bromoethan-1-one] Request for quotation
CAS: 4072-67-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 4072-67-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 4,4'-Bis(2-bromoacetyl)biphenyl Visit Supplier Webpage Request for quotationCAS: 4072-67-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 4,4'-Bis(2-bromoacetyl)biphenyl Visit Supplier Webpage Request for quotationCAS: 4072-67-7
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 1,1'-([1,1'-Biphenyl]-4,4'-diyl)bis(2-bromoethanone) Visit Supplier Webpage Request for quotationCAS: 4072-67-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4,4'-Bis(2-bromoacetyl)biphenyl Request for quotation
CAS: 4072-67-7
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 4072-67-7
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: Isavuconazole Impurity 128 Request for quotation
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: 1,1'-[1,1'-biphenyl]-4,4'-diylbis[2-bromoethan-1-one] Request for quotation
CAS: 4072-67-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 4072-67-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 4,4'-Bis(2-bromoacetyl)biphenyl Visit Supplier Webpage Request for quotationCAS: 4072-67-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
Einecs 223-785-3
252894-39-6
2,3,4,5,6-pentafluoro-benzenemethano
887408-31-3
1,2-BIS(HYDROXYMETHYL)-4-FLUOROBENZENE
Benzoic acid,2-(2-propen-1-ylthio)-
常春藤苷 C
1344545-36-3
1H-Pyrrole-2-carboxaldehyde, 4-acetyl-1,5-dimethyl- (9CI)
(2R,4R)-4-METHYLPIPERIDINE-2-CARBOXYLIC ACID
252894-39-6
2,3,4,5,6-pentafluoro-benzenemethano
887408-31-3
1,2-BIS(HYDROXYMETHYL)-4-FLUOROBENZENE
Benzoic acid,2-(2-propen-1-ylthio)-
常春藤苷 C
1344545-36-3
1H-Pyrrole-2-carboxaldehyde, 4-acetyl-1,5-dimethyl- (9CI)
(2R,4R)-4-METHYLPIPERIDINE-2-CARBOXYLIC ACID





